What are the 8 parts of the periodic table?
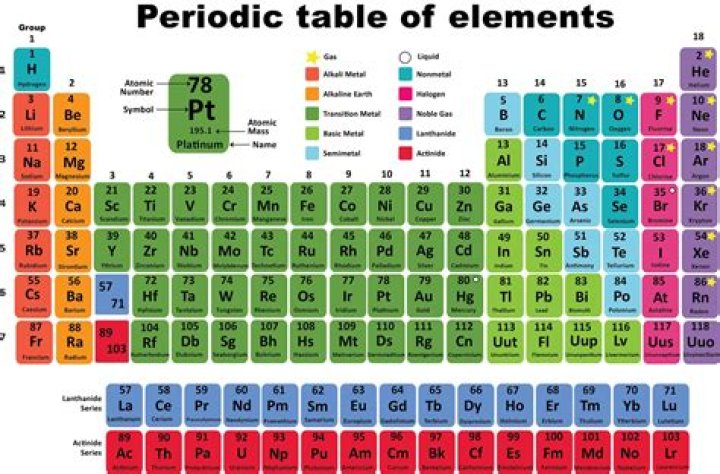
What are the 8 parts of the periodic table?
They are the alkali metals, alkaline earth metals, transition metals, other metals, metalloids, non-metals, halogens, noble gases and rare earth elements.
Are there 8 periods in the periodic table?
There are seven periods in the periodic table, with each one beginning at the far left. A new period begins when a new principal energy level begins filling with electrons. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. Periods 4 and 5 have 18 elements.
Where is 8A on the periodic table?
Group 8A — The Noble or Inert Gases. Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). The name comes from the fact that these elements are virtually unreactive towards other elements or compounds.
How many elements are there in the periodic table of 2021?
118 elements
This list contains the 118 elements of chemistry. For chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. The first chemical element is Hydrogen and the last is Ununoctium.
What are the 4 parts of the periodic table?
There are four main groups of elements in the periodic table: main group metals, non-metals, metalloids, and transition metals (See types of elements).
Is Element 119 possible?
Ununennium, also known as eka-francium or element 119, is the hypothetical chemical element with symbol Uue and atomic number 119. It is the lightest element that has not yet been synthesized.
What is group and period in periodic table?
The columns of the periodic table are called groups. Members of the same group in the table have the same number of electrons in the outermost shells of their atoms and form bonds of the same type. The horizontal rows are called periods.
What is the other name for Group 8 elements?
Group 8 elements are also known as the noble gasses.
What are the first 10 elements?
What are the First 10 Elements
| Name of the Element | Symbol of the Element | Atomic Number |
|---|---|---|
| Carbon | C | 6 |
| Nitrogen | N | 7 |
| Oxygen | O | 8 |
| Fluorine | F | 9 |
Has element 119 been made?
When was the eighth period added to the periodic table?
As some superheavy elements were predicted to lie beyond the seven-period periodic table, an additional eighth period containing these elements was first proposed by Glenn T. Seaborg in 1969.
What are the seven rows of the periodic table called?
The seven rows of the table, called periods, generally have metals on the left and non-metals on the right. The columns, called groups, contain elements with similar chemical behaviours.
Why is there no consensus on periodic table beyond 120?
As a result of uncertainty and variability in predictions of chemical and physical properties of elements beyond 120, there is currently no consensus on their placement in the extended periodic table.
Is there an interactive version of the periodic table?
There is an interactive periodic table that also illustrates the filling sequence well if you click through the atoms. Elements within a column generally have similar properties, but in some places elements side by side can also be similar.
As some superheavy elements were predicted to lie beyond the seven-period periodic table, an additional eighth period containing these elements was first proposed by Glenn T. Seaborg in 1969.
The seven rows of the table, called periods, generally have metals on the left and non-metals on the right. The columns, called groups, contain elements with similar chemical behaviours.
As a result of uncertainty and variability in predictions of chemical and physical properties of elements beyond 120, there is currently no consensus on their placement in the extended periodic table.
There is an interactive periodic table that also illustrates the filling sequence well if you click through the atoms. Elements within a column generally have similar properties, but in some places elements side by side can also be similar.



